Copper Roof and Aluminum Eavestrough

The selection of Eavestrough material must account for the potential of Galvanic or Electrolytic Corrosion at contact points with other metals and at supports. Also, the special selection is required for corrosive environments such as coastal or polluted industrial atmospheres. Galvanic or Electrolytic Corrosion can occur when dissimilar metals are in contact in the presence of an electrolyte. An electrolyte is any non-metal substance that will conduct an electric current, especially a liquid solution. Water, particularly rainwater, is a good electrolyte.
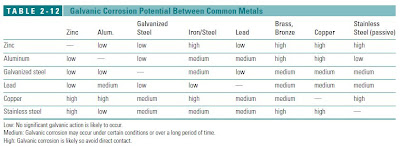
A galvanic scale has been developed for metals. Metals that are more electro-positive (anodize) will corrode more easily and metals that are more electro-negative (cathodic) are more corrosion resistant. When two dissimilar metals are in contact in the presence of an electrolyte, the electro-positive metal will corrode. The farther the two metals are from each other on the galvanic scale, the speed and effect of the reaction will increase.
GALVANIC SCALE:

To reduce the tendency for galvanic corrosion to occur between dissimilar metals one or more of the following
actions can be taken:
1. Avoid contact between metals that are farther apart on the scale.
2. Do not couple a small exposed area of a more electro-positive (anodize) metal with a large area of a more
electro-positive (cathodic) metal. Fasteners are a particular concern in this regard.
3. Coat the electro-positive (cathodic) metal with a suitable paint or another non-metallic coating (or coat both
surfaces at their interface). Zinc chromate or bituminous coatings are often used.
4. Separate the metals by tape, gasket, waterproof paper, elastomeric sheet, sealant or other non-absorbent,
non-conductive material.
5. Do not allow moisture runoff from a more electro-negative (cathodic) metal to drain on a more electro-positive
(anodize) metal, even if the metals are not in contact.
6. Remove metal particles that are deposited from steel dies on formed metals (or prevent the deposits from
occurring).
Miscellaneous Information about Galvanic Corrosion:
1. Passivized stainless steel has been chemically cleaned, usually with an acid solution.
2. Anodized aluminum is considered compatible with all other metals.
3. Salts increase the electrical conductivity of water.
4. The discharge from drain pipes on rooftop air-conditioning units can be very corrosive, particularly if the
piping is copper and the drainage goes into galvanized steel scuppers, conductor heads or gutters.
5. Copper nails/fasteners should not be used for fastening galvanized steel roof panels even though the contact area of the more electro-positive (anodize) zinc coating is small compared to the more electron-negative
(cathodic) copper metal. Galvanized steel nails/fasteners will corrode rapidly if used on copper roofing.
6. Many organizations and individuals have published corrosion tables. All of them tend to rank corrosion rates in non-quantitative terms such as mild, moderate and severe. Rates of corrosion are also affected by conditions such as seacoast, dry inland, acid rain and polluted industrial atmospheres.
Question
I have a porch and bay roof that we plan to replace with copper and because the
trim is white below the copper roof, we were going to put on white aluminum
eavestrough to go with the porch. Some vendors suggest that corrosion will occur,
eventually. Other people have suggested that it shouldn’t be a problem. Do you
have any thoughts about this approach?
Answer
Whenever 2 dissimilar metals come in contact with each other, while in the presence of an electrolyte, a galvanic reaction will occur. The metal that is higher on the nobility scale (Copper) will attractions from the other metal (Aluminum), causing it to corrode or dissolve.
Water is an excellent electrolyte, but just the moisture in the air is enough to get the reaction started, once it starts, you can’t really stop it.
If rainwater runs across Copper and into an Aluminum eavestrough, the electrolyte (water) will attack the Aluminum. The Aluminum will stain and start corroding, but there are too many variables to be able to offer you a timeline. It could last a year or 10 years.
The same would happen with galvanized steel eavestrough.
So you have a choice of Copper, Copper clad stainless, terne-coated stainless, bare stainless or some kind of plastic.
Regards
GTA local Eavestrough | Gutter professionals – Maxima Aluminum LTD



